|
Simple
Vaporizers
These vaporizers
consist of a vaporizing chamber, usually a glass bowl or bottle,
containing liquid anesthetic and a tap controlling the proportion
of gas flowing through it. Examples include the Ohio and Boyle's
bottles, and the Stephens and Goldman vaporizers.
 |
 |
| Boyle's
bottle |
Ohio
8 Bottle |
 |
 |
| Stephens
Universal Vaporizer |
Goldman
vaporizer |
The vaporizing
chamber may contain a wick to improve vaporization. Nonetheless,
the gas in the chamber is unlikely to be fully saturated with
anesthetic. The concentration leaving the vaporizing chamber
is, therefore, less than the saturated vapor pressure and is
usually unknown. The concentration of anesthetic emerging from
the vaporizer is, therefore, also not known. Also, higher gas
flow rates will tend to lower the concentration further since
the carrier gas spends less time in the vaporizing chamber and
less anesthetic is vaporized.
Changes
in temperature
No compensation is made for the effect of changes in temperature
on SVP, so the output of such vaporizers is highly dependent
upon the ambient temperature:
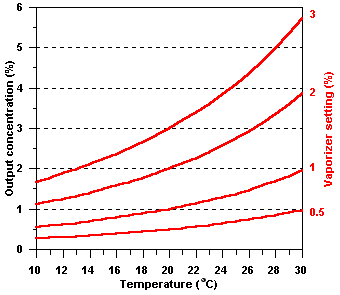 |
| Effect
of changes in temperature on the output concentration of
a simple halothane vaporizer calibrated at 20 oC. |
Time
The
delivered concentration will also be reduced over time, since
loss of the latent heat of vaporization causes the liquid to
cool and lowers the saturated vapor pressure. This is most noticeable
in the case of ether vaporizers, where ice may form on the outside
of the vaporization chamber due to the extreme cold which develops
owing to evaporation.
Accuracy
Because of these causes of variability in their output, simple vaporizers
cannot generally be provided with a scale indicating the concentration
of anesthetic being delivered. Most merely have some qualitative
scale such as Off to On, or 0 to 10. Adjustments to the vaporizer
can only be made on the basis of the response of the patient as
judged by changes in the plane of anesthesia.
Uses
The major advantage of simple vaporizers is their relatively low
cost. Many are also designed to have a low internal resistance which
allows them to be used as draw-over vaporizers.
Although
simple vaporizers may in principle be used to vaporize any agent,
the large difference in the saturated vapor pressure of different
anesthetics presents some practical difficulties. For example, vaporizers
intended for use with halothane or isoflurane are designed to be
relatively inefficient in order to produce a maximum output of around
3%. If such vaporizers were to be used to vaporize methoxyflurane,
it would be difficult to produce an adequate concentration due to
the very much lower SVP of this agent. Conversely, were a methoxyflurane
vaporizer (such as the Ohio bottle) used to vaporize isoflurane,
lethally high concentrations might be produced.
The
disadvantage of simple vaporizers is their unknown and variable
output. This does not present a major problem when slowly-equilibrating
agents (such as methoxyflurane or ether) are used and reasonable
vigilance is employed. However, the rapid changes in depth of anesthesia
which may be achieved with halothane and isoflurane may cause major
difficulties to occur if their concentration is not known.
Precision
vaporizers 
|